Correct option is C
The correct answer is (c) ~10 μmol L⁻¹.
Understanding Alkalinity:
· Alkalinity measures the water's capacity to neutralize acid or the concentration of bases in water.
· In this case, the water sample has a pH of 9 and no carbonate or other proton donors/acceptors, meaning alkalinity is contributed primarily by hydroxide ions (OH⁻).
Calculation of Hydroxide Ion Concentration:
Given the pH of the water sample is 9:
1. pH = 9
· This means the concentration of H⁺ ions is:
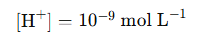
2. Since water dissociation produces equal amounts of H⁺ and OH⁻, the concentration of OH⁻ ions can be calculated using the water dissociation constant (Kw):
3. Therefore:
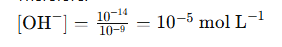
4. Converting to μmol L⁻¹:
10−5 mol L−1 equivalent to 10 μmol L−1.
Information Booster 1. Alkalinity Sources:
· Typically comes from carbonate (CO₃²⁻), bicarbonate (HCO₃⁻), and hydroxide (OH⁻) ions.
2. pH and Alkalinity Relationship:
· pH 7: Neutral water (equal H⁺ and OH⁻).
· pH > 7: Basic/alkaline water (higher OH⁻ concentration).
· pH < 7: Acidic water (higher H⁺ concentration).
3. Units of Alkalinity:
· Often expressed in mg/L as CaCO₃ or in mol/L.
Additional Knowledge · Importance of Alkalinity:
· Maintains pH stability in water systems.
· Essential for the health of aquatic life and the efficiency of water treatment processes.
· Measurement of Alkalinity:
· Measured via titration using a strong acid (e.g., HCl) until a specific endpoint pH (commonly pH 4.5) is reached.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins


