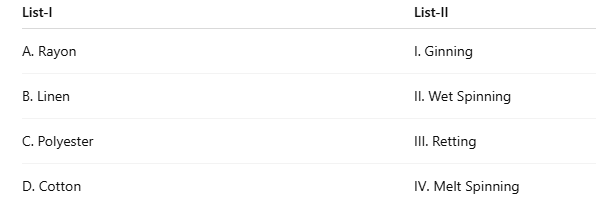
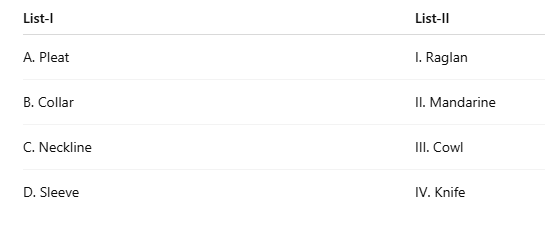
Correct option is B
The ion described in the question is called a
Zwitterion. A
zwitterion is a molecule that has
both positive and negative charges but is
electrically neutral overall. At the
isoelectric point, an amino acid or protein can exist as a zwitterion, where the
amine group is protonated and carries a
positive charge, while the
carboxyl group is deprotonated and carries a
negative charge. The
net charge is zero at this point.
Information Booster: The
isoelectric point (pI) of an amino acid or peptide is the pH at which the molecule has no net charge and exists in the zwitterionic form. This is important in various biochemical processes, including protein solubility and electrophoresis.
Additional Knowledge:
·
Isoelectric Ions: This term is not commonly used in biochemistry;
zwitterions are more commonly discussed.
·
Zero Ions: This term does not accurately describe the phenomenon related to isoelectric point or zwitterions.
·
Ionizable Ions: This is a general term for ions that can either donate or accept protons, but does not specifically refer to the condition of having both positive and negative charges simultaneously, as in the case of zwitterions.
Top of Form





 English
English 150 Questions
150 Questions 150 Mins
150 Mins