Correct option is C
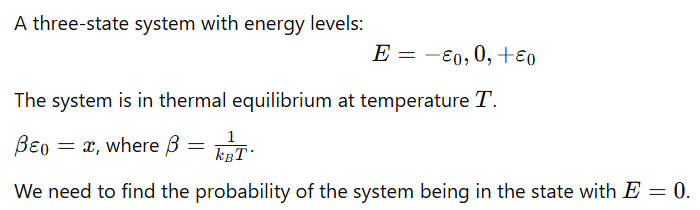
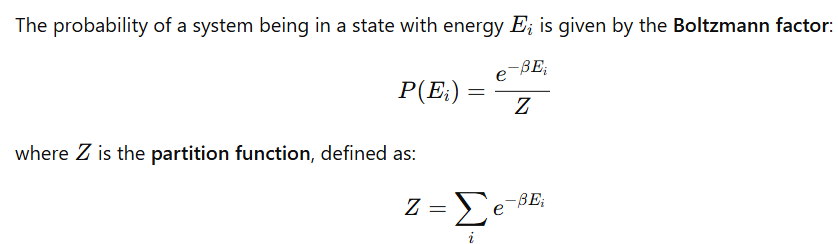

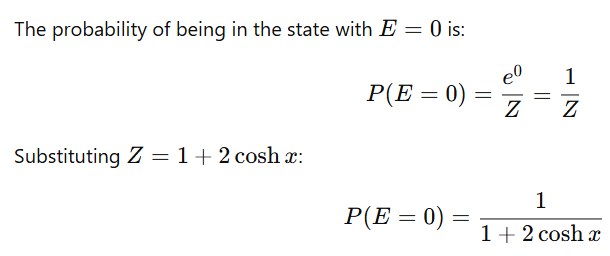

A three-state system with energies
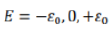
is in a thermal equilibrium at a temperature T.
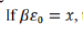
the probability of finding the system with energy E=0 is
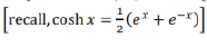
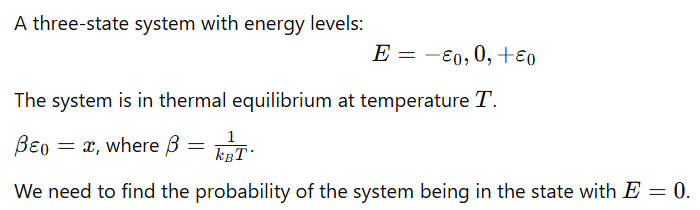
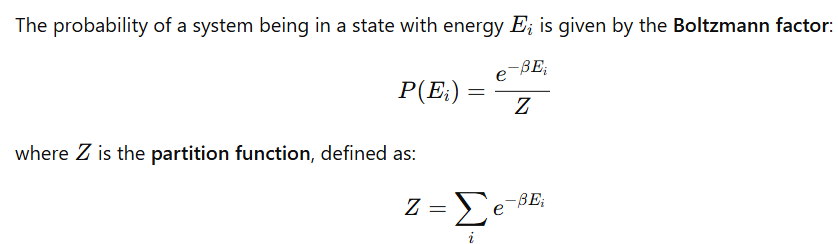

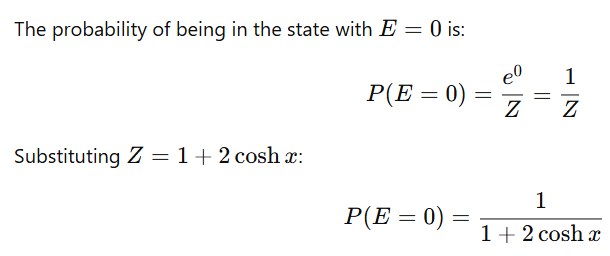

The system with the largest internal pressure is
A compound consists of atoms A and B. Atoms of B form cubic close packing lattice and atoms of A occupy half of octahedral voids and half of tetrahedral voids. What is the molecular formula of the compound?
Entropy associated with a die with all its six faces having equal probability of being on top is (kB : Boltzmann constant):

The equivalent symmetry operations for and are, respectively,
The system with the largest internal pressure is
A compound consists of atoms A and B. Atoms of B form cubic close packing lattice and atoms of A occupy half of octahedral voids and half of tetrahedral voids. What is the molecular formula of the compound?
Entropy associated with a die with all its six faces having equal probability of being on top is (kB : Boltzmann constant):

The equivalent symmetry operations for and are, respectively,

