Correct option is C
The correct answer is: (c) Substitution reaction
- A substitution reaction occurs when one atom or a group of atoms in a molecule is replaced (or substituted) by another atom or group of atoms, leading to the formation of a new compound.
Example of a Substitution Reaction:
In the reaction between methane (CH₄) and chlorine (Cl₂) under the influence of light:
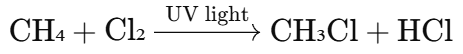
Here, one hydrogen atom in methane is replaced by a chlorine atom.
Information Booster
Oxidation:
· Oxidation is a reaction where a substance loses electrons or gains oxygen.
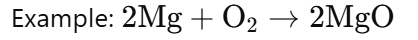
· This does not involve replacement of atoms, so it is not a substitution reaction.
Addition Reaction:
· In an addition reaction, two or more reactants combine to form a single product.
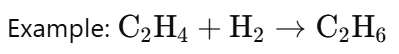
· There is no replacement of atoms, so this is not correct.
Combustion:
· Combustion is a reaction where a substance reacts with oxygen to produce heat and light.
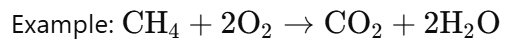
· This is an oxidation-reduction process, not a substitution.





 English
English 100 Questions
100 Questions 60 Mins
60 Mins

