Correct option is D
Solution:
From the first law of thermodynamics:
Start with the general form of the first law:
dQ = dU + p dVSubstitute specific heat terms:
dQ = Cv dT + p dV
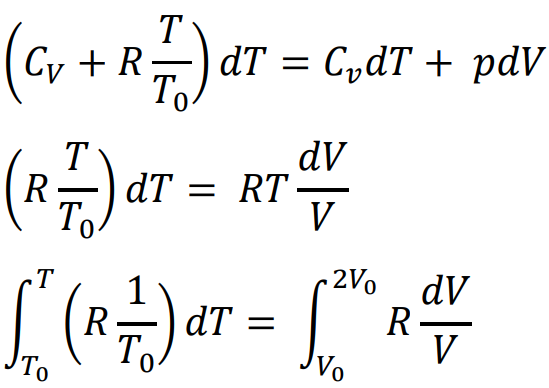
A classical ideal gas is subjected to a reversible process in which its molar specific heat changes with temperature T as . If the initial temperature and volume are T0 and V0, respectively, and the final volume is 2V0, then the final temperature is
Solution:
From the first law of thermodynamics:
Start with the general form of the first law:
dQ = dU + p dVSubstitute specific heat terms:
dQ = Cv dT + p dV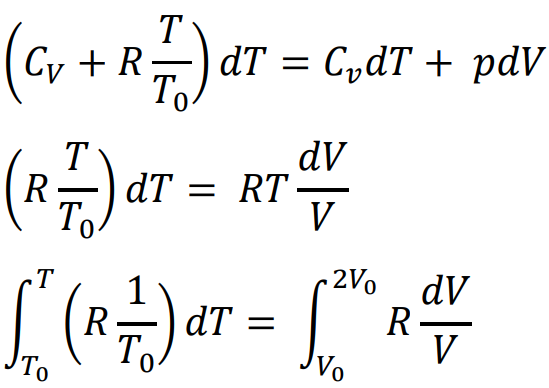
The following ternary figure shows the marks obtained by students A, B and C in an exam of 100 marks. Here, C received 10 marks in Physics, 50 marks in Chemistry and 40 marks in Mathematics.
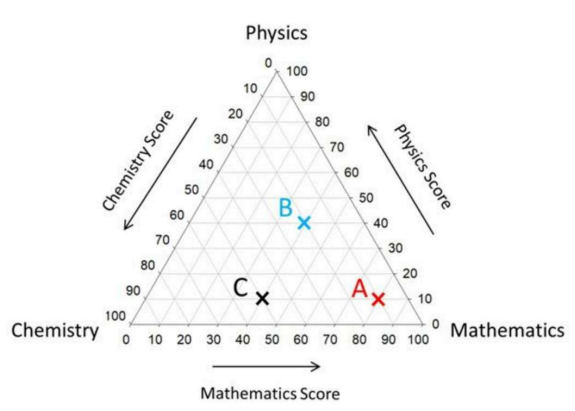
Which one of the following is CORRECT?
The Sankey diagram shows food preferences of males and females. The widths of the bands are proportional to the number of persons opting for a given food item.
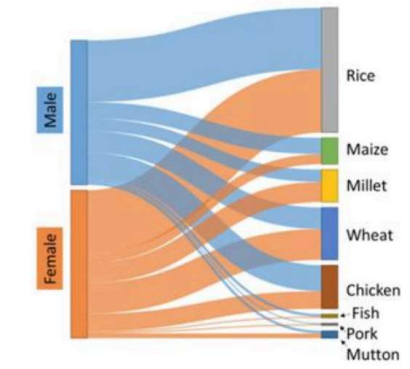
Which one of the following is an INCORRECT statement about the food preferences?
Consider the following information regarding a 3-digit PIN.

The correct PIN is
With a certain set of diameters, a tractor’s front wheels make 2 revolutions for every 1 revolution of the rear wheels. The effect of changing wheel diameter(s) is shown in the given plot.
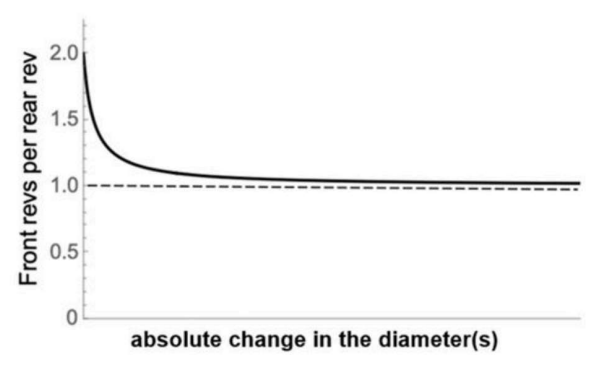
In this context, which of the following statements is CORRECT?
The pie charts show placement percentages of graduates of five different subjects out of the total campus placements for two consecutive years (2023 and 2024).
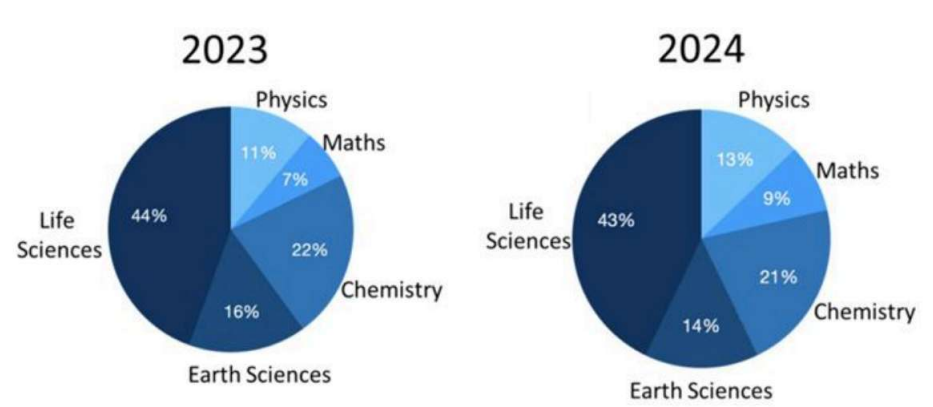
In the year 2024, the number of Maths students who got placed was double that of the previous year. Which of the following is closest to the percentage change in the number of Life Sciences students placed in 2024?

