Correct option is C
The correct answer is (c) hydration to yield another hydrate
Plaster of Paris (POP) is chemically known as calcium sulfate hemihydrate (CaSO4⋅1/2H2O).
When mixed with water, it undergoes a hydration process to form calcium sulfate dihydrate (gypsum) (CaSO4⋅2H2O).
Chemical Reaction:
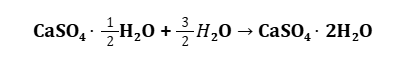
This reaction releases heat (exothermic) and causes the plaster to set into a hard mass.
Additional Information:
- Dehydration: Dehydration is the removal of water, which occurs during the preparation of Plaster of Paris from gypsum, not during its setting.
- Oxidation with atmospheric oxygen: There is no reaction involving oxygen during the setting of Plaster of Paris.
- Combination with atmosphere: The setting process does not involve atmospheric components like oxygen or carbon dioxide.





 English
English 40 Questions
40 Questions 30 Mins
30 Mins are:
are:


