Correct option is C
When a gas is dissolved in a solid, it forms a solid solution. In the case of hydrogen and palladium, hydrogen gas molecules (H2) are absorbed into the crystal lattice of the palladium metal.
The hydrogen atoms occupy interstitial sites in palladium hydride. The H–H bond in H2 is cleaved. The ratio in which H is absorbed on Pd is defined by
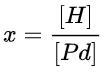 x=[H][Pd] When Pd is brought into a H2 environment with a pressure of 1 atm, the resulting concentration of H reaches
x ≈ 0.7.
x=[H][Pd] When Pd is brought into a H2 environment with a pressure of 1 atm, the resulting concentration of H reaches
x ≈ 0.7.
Additional Knowledge:
· Camphor in nitrogen gas: This is an example of a gas-gas solution where camphor sublimates into a gaseous phase and mixes with nitrogen.
· Oxygen dissolved in water: This represents a liquid solution where oxygen molecules dissolve into water. This is commonly seen in aquatic ecosystems where dissolved oxygen supports marine life.





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

