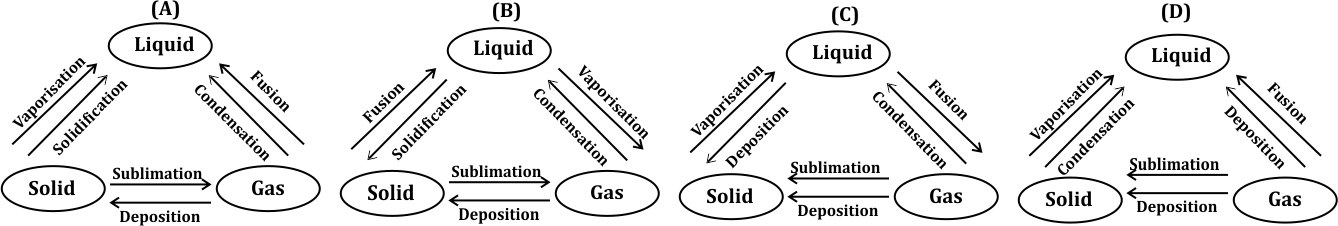
Correct option is D
The correct answer is (d) B.
Option (B) accurately represents the interconversion of the three states of matter (solid, liquid, and gas) with all processes correctly labeled. This diagram is complete and accurate, showcasing all the necessary processes for transitioning between each state of matter.
Information Booster:
· Melting is the process of changing a solid into a liquid by heating.
· Fusion occurs when two light atomic nuclei collide and combine to form a heavier nucleus, releasing a large amount of energy. The process of converting a solid into its liquid state is called fusion.
· Vaporization occurs when a substance changes from a liquid or solid state to a gaseous state.
· Condensation happens when water vapor in the air turns into liquid water and forms droplets on a cold surface.
· Solidification is the process of a liquid changing into a solid, which happens when the temperature of the liquid is lowered below its freezing point.
· Sublimation is the transition of a solid directly into a gas without passing through the liquid phase.
· Deposition is the reverse of sublimation, where a gas changes directly into a solid.





 English
English 100 Questions
100 Questions 60 Mins
60 Mins

