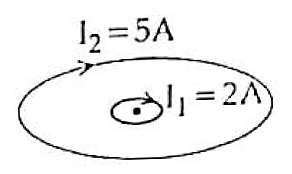Correct option is C
The correct answer is: (c) Blue
Explanation: The transition from n = 4 to n = 2 in a hydrogen atom falls under the Balmer series, which lies in the visible region of the electromagnetic spectrum. This specific transition emits blue light with a wavelength of approximately 486 nm.
Information Booster:
· The Balmer series involves transitions that end at the second energy level (n = 2).
· Transitions from higher energy levels (n > 2) to n = 2 produce visible light of different colors.
· The color depends on the energy gap; for n = 4 to n = 2, it corresponds to blue.





 English
English 150 Questions
150 Questions 150 Mins
150 Mins