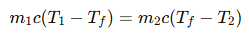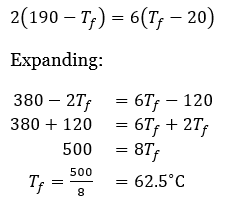Correct option is C
To determine the final equilibrium temperature, we apply the
principle of conservation of energy, which states that the heat lost by the hot water is equal to the heat gained by the cold water.
Heat lost = Heat gained
Since we assume
no heat loss to the surroundings, we use the formula: