Correct option is B
Ans.(b)
For zero order reaction
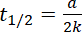
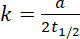
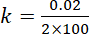
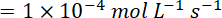
Ans.(b)
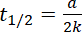
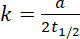
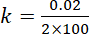
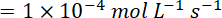
 English
English 200 Questions
200 Questions 200 Mins
200 Mins English
English English
English 200 Questions
200 Questions 200 Mins
200 Mins English
English
 the rate of reaction will :
the rate of reaction will :
 .
.
 represents
represents
 is consistent with Arrhenius equation?
is consistent with Arrhenius equation?
 Calculate the energy of activation.
Calculate the energy of activation.

Suggested Test Series




Suggested Test Series



