Correct option is B
Ans.(b)
Rate of reaction according to collision theory can be expressed as
Rate =
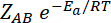
Where, ZAB represents the collision frequency of the reactants, A & B and
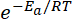
represents the fraction of molecules with energies equal to or greater than Ea
The number of collisions per second per unit volume of the reaction mixture (A and B) is known as collision frequency ZAB.





 represents
represents


 English
English 200 Questions
200 Questions 200 Mins
200 Mins
 the rate of reaction will :
the rate of reaction will :
 .
.
 is consistent with Arrhenius equation?
is consistent with Arrhenius equation?
 Calculate the energy of activation.
Calculate the energy of activation.

