Correct option is C
The correct answer is (c) Z.
· The atomic number of an element is denoted by the letter Z.
· The atomic number represents the number of protons in the nucleus of an atom of a particular element, and it determines the element's identity and position in the periodic table.
For example, in the notation of an element, such as:
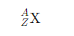
· Z represents the atomic number (number of protons).
· A represents the mass number (number of protons + neutrons).
· X is the chemical symbol for the element.
Additional Knowledge:
· A: This represents the mass number, which is the sum of protons and neutrons in an atom's nucleus.
· X: This typically represents the symbol of the element in chemical notation.
· N: This sometimes refers to the number of neutrons in an atom's nucleus but is not used for the atomic number.





 English
English 100 Questions
100 Questions 90 Mins
90 Mins

