Correct option is B
The correct answer is (b) Grey.
· Silver chloride (AgCl) turns grey in sunlight due to a photochemical decomposition reaction. When exposed to sunlight, silver chloride breaks down into silver metal and chlorine gas:
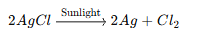
· The silver metal appears as a grey precipitate.
Explanation of Other Options:
· Orange : No orange coloration occurs during the decomposition of silver chloride.
· Red : Red is not related to this reaction.
· White : Silver chloride is originally white in color before exposure to sunlight.
Key Fact: This reaction demonstrates the photo-sensitivity of silver chloride and is the principle behind its use in old photographic films and plates.





 English
English 100 Questions
100 Questions 90 Mins
90 Mins

