Correct option is B
The correct answer is (b) Ethene.
· On heating ethanol (C2H5OH) with concentrated sulfuric acid (H2SO4) at 443 K, ethene (C2H4) is produced. This reaction is known as dehydration of ethanol, where water is removed from the ethanol molecule to form ethene.
Reaction:
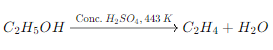
Explanation of Other Options:
· Ethane : Not formed in this reaction; requires hydrogenation of ethene.
· Ethyne : Formed from calcium carbide or other reactions, not directly from ethanol dehydration.
· Methane : Not related to the dehydration of ethanol.





 English
English 100 Questions
100 Questions 90 Mins
90 Mins

