Correct option is D
1.
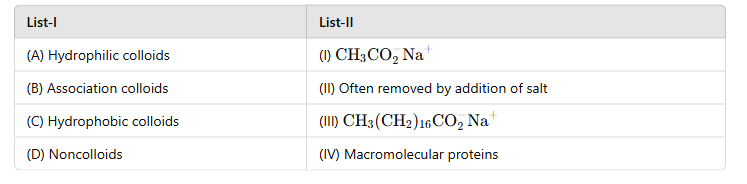
(A) Hydrophilic Colloids → (IV): Macromolecular Proteins
· Hydrophilic colloids are those where particles are well solvated (water-loving). Macromolecular proteins such as albumin are typical examples of hydrophilic colloids.
2.
(B) Association Colloids → (II): Often Removed by Addition of Salt
· Association colloids (or micelles) form when amphiphilic molecules (e.g., soaps) aggregate above a critical micelle concentration. These are often destabilized and removed by the addition of salts.
3.
(C) Hydrophobic Colloids → (III): CH3 (CH2)16CO2−Na+
· Hydrophobic colloids are formed by nonpolar molecules that are water-insoluble. Sodium stearate (CH3 (CH2)16CO2−Na+) is a classic example.
4.
(D) Noncolloids → (I): CH3CO2−Na+
· Noncolloids like sodium acetate (
CH3CO2−Na+) dissolve completely in water and do not form colloidal solutions.
Information Booster: 1. Colloidal Systems: Comprise two phases – dispersed phase and dispersion medium.
2. Hydrophilic Colloids: Stabilized by strong interactions with the solvent. Example: Proteins.
3. Hydrophobic Colloids: Need stabilizers to remain dispersed. Example: Sodium stearate.
4. Association Colloids: Aggregates of amphiphilic molecules, forming micelles.
5. Noncolloids: Substances that dissolve uniformly without forming dispersed systems





 English
English 10 Questions
10 Questions 12 Mins
12 Mins

