Correct option is C
Pure silicon has a crystalline structure where each silicon atom shares its four valence electrons with four neighboring silicon atoms, forming covalent bonds.
To create an n-type semiconductor, a pentavalent element (one with five valence electrons) is added as a dopant. This element has one extra valence electron compared to silicon.
When a pentavalent element such as arsenic is introduced into the silicon lattice, four of its electrons form covalent bonds with neighboring silicon atoms, while the fifth electron remains free.
This free electron increases the conductivity of the silicon by contributing to the flow of electric current, thereby creating an n-type semiconductor.
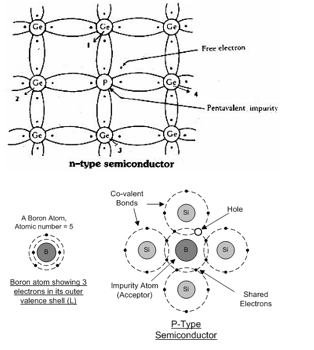
Dopants and Their Effects: (a) Gallium: - Gallium is a trivalent element (with three valence electrons). When added to silicon, it creates a p-type semiconductor by accepting electrons and creating "holes" (places where an electron is missing).
(b) Indium: - Indium is also a trivalent element, similar to gallium, and results in a p-type semiconductor when added to silicon.
(c) Arsenic: - Arsenic is a pentavalent element. When added to silicon, it donates an extra electron, resulting in an n-type semiconductor.
(d) Aluminium: - Aluminium is a trivalent element, which also results in a p-type semiconductor when added to silicon.
Therefore, the correct answer is (c) arsenic






