Correct option is D
Ans.(d)
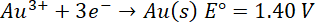
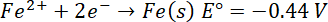
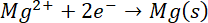
E°=-2.36 V
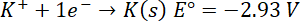
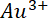
occupies the top position in the electrochemical series.
Ans.(d)
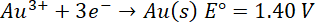
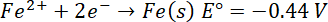
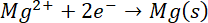
E°=-2.36 V
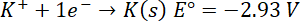
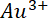
occupies the top position in the electrochemical series.
 English
English 200 Questions
200 Questions 200 Mins
200 Mins English
English English
English 200 Questions
200 Questions 200 Mins
200 Mins English
EnglishThe number of moles of hydrogen oxidised is:
Which of the following best describes why ionic compounds like potassium bromide (KBr) do NOT conduct electricity in solid form but do in molten form?
Which of the following electrolytes is used in the electrolyte refining of copper?



 from water in the presence of an acid?
from water in the presence of an acid?
Suggested Test Series




Suggested Test Series



