Correct option is C
Definition of Electron-Volt (eV)
An electron-volt is defined as the amount of kinetic energy gained or lost by an electron when it is accelerated through an electric potential difference of one volt.
1 eV equal to the exact value 1.6 ×10−19 J.
The electron-volt is a convenient unit of energy often used in atomic, nuclear, and particle physics.
It is especially useful in these fields because the energy changes involved are typically on the scale of single electrons and very small energy quantities.
eV and Joule are the units of energy in two different systems. Their equivalence can be mathematically expressed as-
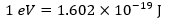
Where,
eV is electron volt
J is Joule
Electron-Volt: -
1 electron volt is the energy change that takes place when a charge equal to 1 electron (1.6×10-19 C) is moved through a potential difference of 1 volt.
1eV = 1.602×10-19 J
Symbol: - eV
Joule: -
1 Joule is the work done or energy transferred on an object when a force of 1 newton acts on it in the direction of its motion along a distance of 1 meter.
1 J = 6.2415×1018 eV
Symbol: - J
Detailed Solution
(a) Electric Potential: Measured in volts (V), it represents the potential energy per unit charge.
(b) Electric Power: Measured in watts (W), it represents the rate at which electrical energy is transferred or converted.
(c) Electric Energy: Measured in joules (J) in the SI system, or electron-volts (eV) in atomic and subatomic contexts.
(d) Electric Force: Measured in newtons (N), it represents the force between two charges.






