Correct option is B
Explanation-
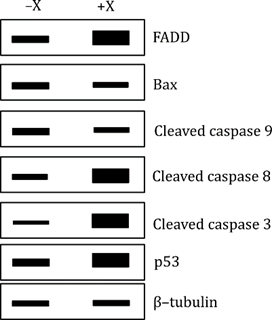
Cervical cancer cells were either left untreated (−X) or treated with a compound ‘X’ (+X), which is being tested as an anti-cancer drug. Researchers used immunoblotting (Western blotting) to detect the levels of different proteins that are involved in cell death (apoptosis). The thickness of the bands in the blot tells us how much of each protein is present.
FADD levels increased in treated cells (+X) - FADD is a key adaptor molecule in the extrinsic pathway of apoptosis, which is triggered from outside the cell (typically through death receptors like Fas). So, this suggests that compound ‘X’ is activating the extrinsic apoptosis pathway.
Cleaved caspase 8 levels increased with treatment - Caspase 8 is activated downstream of FADD in the extrinsic pathway. Its cleavage confirms that the extrinsic apoptotic pathway is indeed being activated.
Cleaved caspase 3 also increased - Caspase 3 is a common executioner caspase in both extrinsic and intrinsic pathways. So, increased cleaved caspase 3 shows that apoptosis is happening.
p53 levels increased- p53 is a tumor suppressor that promotes apoptosis in response to DNA damage or stress. Its increase suggests that the apoptosis induced by compound X is at least partially dependent on p53.
Bax and cleaved caspase 9 showed no significant change - These are critical components of the intrinsic (mitochondrial) pathway. If the intrinsic pathway were involved, we would expect to see Bax upregulated and cleaved caspase 9 increased. Since that didn’t happen, the intrinsic pathway is not significantly involved.
Conclusion:
FADD ↑ and cleaved caspase 8 ↑ = extrinsic pathway activation
p53 ↑ = apoptosis is p53-dependent
Bax and caspase 9 (intrinsic pathway) show no change, so intrinsic pathway not involved
So, the correct answer is option B - Compound ‘X’ induced cell death via the extrinsic pathway by inducing the Fas ligand-associated death domain (FADD) and apoptosis was p53 dependent.