Correct option is B
Ans.(b)
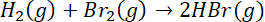
;
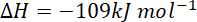
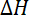
= sum of bond energy of reactants – sum of bond energy of products
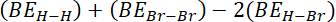
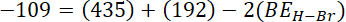


Ans.(b)
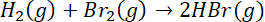
;
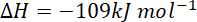
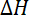
= sum of bond energy of reactants – sum of bond energy of products
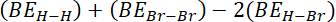
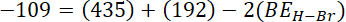

 English
English 200 Questions
200 Questions 200 Mins
200 Mins English
English English
English 200 Questions
200 Questions 200 Mins
200 Mins English
EnglishIdentify the incorrect statement.

 for which the same reaction will be spontaneous at the same temperature is
for which the same reaction will be spontaneous at the same temperature is


 from pressure of 20 atmosphere to 10 atmosphere is:
from pressure of 20 atmosphere to 10 atmosphere is:



 for one mole of ideal gas?
for one mole of ideal gas?
Suggested Test Series




Suggested Test Series



