Correct option is B
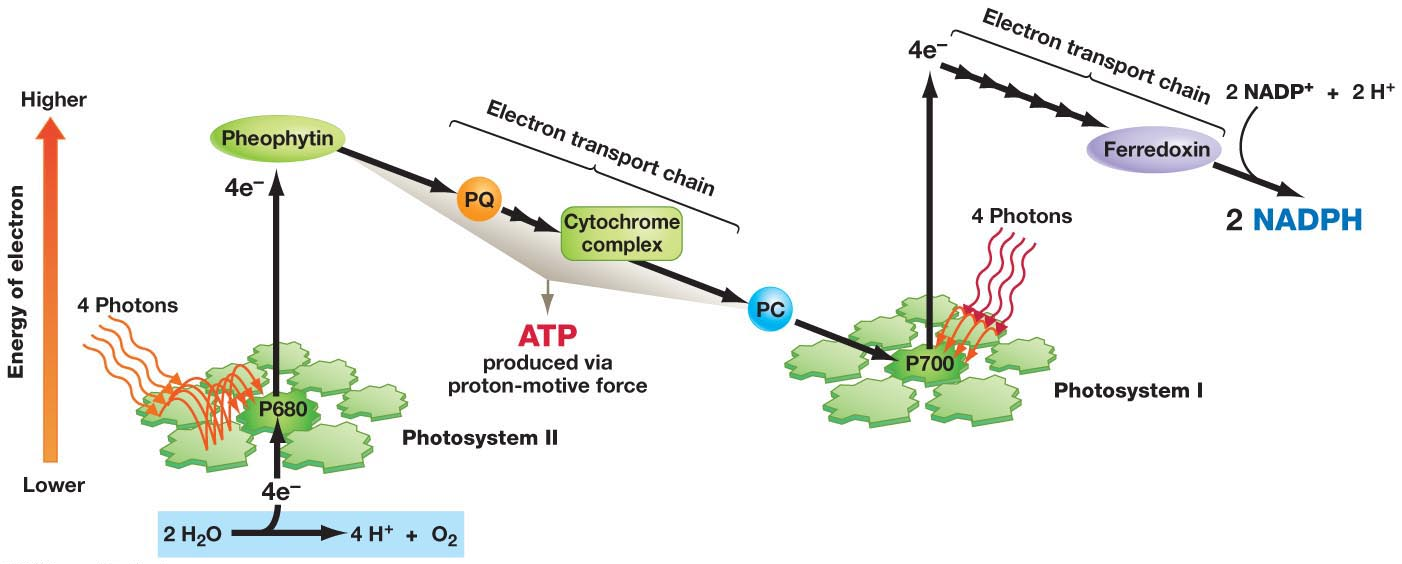
In the process of photosynthesis, the strongest oxidizing agent is P680⁺, which is the oxidized form of P680, the reaction center in photosystem II (PSII). P680⁺ is a very powerful oxidizing agent and is capable of extracting electrons from water molecules, leading to the production of oxygen (O₂) through water splitting.
The P680⁺ state is critical in the light-dependent reactions of photosynthesis because it enables the initial electron transfer that begins the process of photosynthetic electron transport. P680⁺ has a very high affinity for electrons, making it the strongest oxidizing agent produced during photosynthesis.
Explanation of Other Options:
- NADPH: NADPH is a product of the light reactions and is used in the Calvin cycle for carbon fixation. While NADPH is an electron donor, it is not an oxidizing agent.
- Ferredoxin: Ferredoxin is involved in the electron transport chain but is a reducing agent, not an oxidizing agent.
- P700⁺: P700⁺ is the oxidized form of P700, the reaction center of photosystem I (PSI). It is a strong oxidizing agent but not as strong as P680⁺.