Correct option is D
The correct answer is (d) Only III
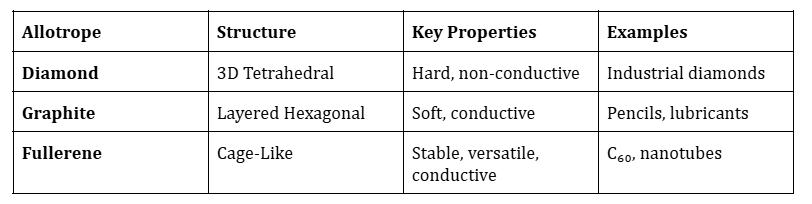
Fullerene:
- Fullerenes are molecules composed entirely of carbon atoms arranged in a cage-like structure.
- The most common fullerene is C₆₀, also known as Buckminsterfullerene, which resembles a soccer ball.
- These molecules have a hollow, spherical, ellipsoidal, or tubular shape.
Diamond:
- Diamond has a tetrahedral crystal lattice structure, not a cage-like structure.
- Each carbon atom is bonded to four other carbon atoms in a 3D network.
Graphite:
- Graphite has a layered structure with carbon atoms arranged in hexagonal sheets, not a cage-like structure.
- These sheets are held together by weak van der Waals forces.
Additional Information:
Why Fullerenes Have Cage-Like Structures:
- Fullerenes form a closed geometry (spherical, tubular, or ellipsoidal), making them unique among carbon allotropes.
- The structure is formed by a combination of pentagons and hexagons, creating a hollow, stable configuration.
Key Differences Between Carbon Allotropes:





 English
English 40 Questions
40 Questions 30 Mins
30 Mins

