Correct option is A
The correct answer is (a) C.
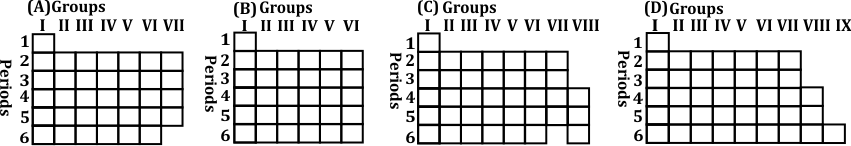
Option (C) accurately represents the structure of Mendeleev's Periodic Table as published in the German journal in 1872. This version of the periodic table organizes elements by increasing atomic weight and groups elements with similar chemical properties in the same vertical columns. Mendeleev’s layout also includes intentional gaps for elements he predicted would be discovered later, and it stops at Group VIII as per his original design.
Information Booster:
● Mendeleev’s Periodic Table was based on atomic weights, which he organized to reflect periodic patterns in properties. ● The vertical columns represent groups of elements with similar chemical behaviors. ● Mendeleev left gaps in his table to account for undiscovered elements, predicting their properties accurately. ● The table became a foundation for the modern periodic table, which organizes elements by atomic numbers. ● Mendeleev’s periodic law stated that the properties of elements are periodic functions of their atomic weights (later revised to atomic numbers). ● His accurate predictions and the periodic arrangement of elements helped validate his table, making it widely accepted in the scientific community.





 English
English 100 Questions
100 Questions 60 Mins
60 Mins

