Correct option is C
1. Concentration of Oxygen Calculation:
· The atmosphere contains approximately 21% oxygen by volume on a dry basis.
· To calculate the concentration of oxygen in g/L, use the Ideal Gas Law:
PV=nRT
Where: P=1 atm, T=273.15 KT, R=0.0821
The molar mass of oxygen (O2) is 32 g/mol.
Molar volume at standard temperature and pressure (STP) for gases:
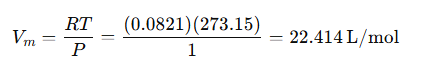
Mass of oxygen per liter:

Substituting:
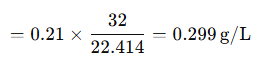
The concentration of oxygen at STP is approximately 0.299 g/L.
Information Booster: 1. Atmospheric Composition by Volume (Dry Air):
· Nitrogen (N2N_2N2): ~78%
· Oxygen (O2O_2O2): ~21%
· Argon (ArArAr): ~0.93%
· Trace Gases (CO₂, Neon, etc.): ~0.07%
2. Oxygen Concentration Variations:
· Varies slightly with altitude and temperature.
· Higher altitudes have lower partial pressures of oxygen.
3. Significance in Environmental Science:
· Oxygen concentration is crucial for combustion, respiration, and industrial applications.
· Monitoring oxygen levels is vital for pollution studies and maintaining air quality.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins

