Correct option is B
Given:
- Red (R) = 4°C
- Green (G) = 8°C
- Blue (B) = 2°C
Concept Used:
The positioning of water layers is determined by density, which is inversely related to temperature (i.e., colder water is denser and sinks).
Thus, the colder the water, the lowerit will appear in the jar.
Explanation:
Densities from highest to lowest (based on temperature):
- Blue (2°C) → Most dense
- Red (4°C) → Medium dense
- Green (8°C) → Least dense
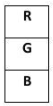
So the natural order from bottom to top is:
- Bottom → Red (4°C)
- Middle → Blue (2°C)
- Top → Green (8°C)
This means Blue (2°C)is in the middle, being denser than Green but lighter than Red — due to unique water behavior near 4°C, where water has maximum density.
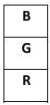
Final Order:
- Top: Green (G, 8°C)
- Middle: Blue (B, 2°C)
- Bottom: Red (R, 4°C)