Correct option is D
Correct answer is D
Explanation:
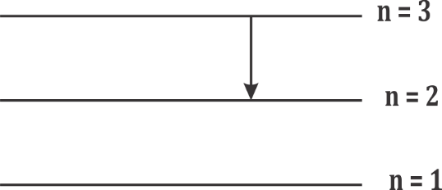
In the hydrogen atom, spectral lines are grouped into different series based on the final energy level (nf) to which the electron transitions:
Lyman series: Transitions to nf = 1
Balmer series: Transitions to nf = 2
Paschen series: Transitions to nf = 3
Brackett series: Transitions to nf = 4
The transition shown occurs to nf = 2. Therefore, it belongs to the Balmer series.





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

