Correct option is B
A zero-order reaction is characterized by a rate that is constant and independent of the concentration of the reactant. The rate law for a zero-order reaction is:
Rate=k
Here, k is the rate constant, and the rate does not depend on the reactant concentration. Thus, the rate remains constant throughout the reaction, irrespective of how much reactant is present. This is why the rate equals the rate constant in zero-order reactions.
Information Booster:
1. Units of k: The units of the rate constant k for zero-order reactions are molL-1s-1.
2. Examples of zero-order reactions: Catalytic decomposition of ammonia and photochemical decomposition of HI in the presence of light.
3. Half-life: The half-life of zero-order reactions depends on the initial concentration:
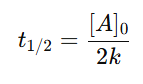
Additional Knowledge:
First Order: First-order reactions depend on the concentration of the reactant, with the rate law given by Rate=k[A]. The rate is not constant and changes with the concentration of the reactant.
Second Order: The rate of a second-order reaction depends on either the square of a single reactant’s concentration (Rate=k[A]2) or the product of two reactants’ concentrations (Rate=k[A][B]). The rate constant k for second-order reactions has units of Lmol-1s-1.





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

