Correct option is C
For strong acids:
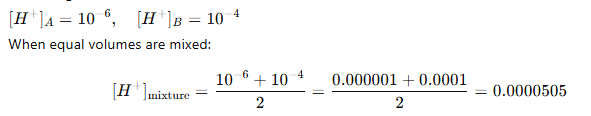
pH = –log(5.05 × 10⁻⁵) ≈ 4.3 So, pH lies
between 4 and 5.
For strong acids:
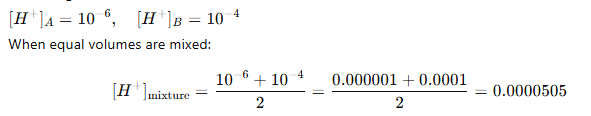
pH = –log(5.05 × 10⁻⁵) ≈ 4.3 So, pH lies
between 4 and 5.
 English
English 100 Questions
100 Questions 120 Mins
120 Mins English
English English
English 60 Questions
60 Questions 45 Mins
45 Mins English
English English
English 30 Questions
30 Questions 27 Mins
27 Mins English
English| LIST I |
LIST II |
|
| A |
Starch and Inulin |
I. Animals |
| B |
Maltose |
II. Very long chain of glucose polymer |
| C |
Cellulose |
III. Plants |
| D |
Glycogen |
IV. Disaccharide of two glucose molecules |

Suggested Test Series






Suggested Test Series




