Correct option is B
There is a large class of reactions, known as 1, 3-dipolar cycloaddition reactions. The species
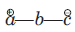
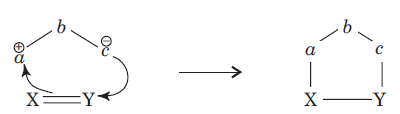
is called the 1, 3-dipolar molecule and X=Y is the dipolarophile.
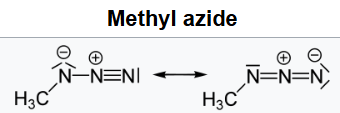
Methyl azide is an organic compound with the formula CH3N3. It is a white solid and it is the simplest organic azide.
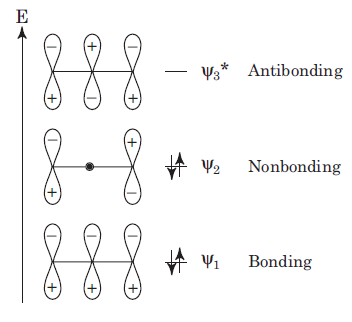
1, 3-Dipolar species has three p-orbitals having four electrons hence HOMO will be ψ2 which has c2-symmetry. The π orbitals of the 1, 3-dipolar species can be represented as follows: