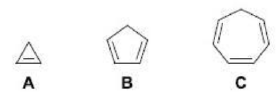
Correct option is B
Aromatic compounds
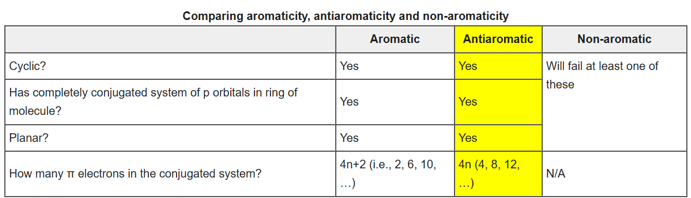
Aromatic compounds or arenes are organic compounds "with a chemistry typified by benzene" and "cyclically conjugated." Aromatic compounds are now defined as cyclic compounds satisfying Hückel's Rule.
In organic chemistry, Hückel's rule predicts that a planar ring molecule will have aromatic properties if it has 4n + 2 π-electrons, where n is a non-negative integer. A cyclic ring molecule follows Hückel's rule when the number of its π-electrons equals 4n + 2, although clearcut examples are really only established for values of n = 0 up to about n = 6.
Criteria for simple aromatics are:
1. the molecule must have 4n + 2 (a so-called "Hückel number") π electrons (2, 6, 10,..) in a conjugated system of p orbitals (usually on sp2-hybridized atoms, but sometimes sp-hybridized);
2. the molecule must be (close to) planar (p orbitals must be roughly parallel and able to interact, implicit in the requirement for conjugation);
3. the molecule must be cyclic (as opposed to linear);
4. the molecule must have a continuous ring of p atomic orbitals (there cannot be any sp3 atoms in the ring, nor do exocyclic p orbitals count).
Antiaromatic compounds
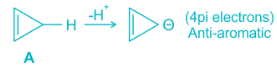
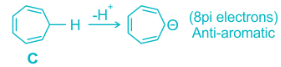
Antiaromaticity is a chemical property of a cyclic molecule with a π electron system that has higher energy, i.e., it is less stable due to the presence of 4n delocalised (π or lone pair) electrons in it, as opposed to aromaticity. Unlike aromatic compounds, which follow Hückel's rule ([4n+2]π electrons) and are highly stable, antiaromatic compounds are highly unstable and highly reactive.
The term 'antiaromaticity' was first proposed as "a situation in which a cyclic delocalization of electrons is destabilising". The IUPAC criteria for antiaromaticity are as follows:
1. The molecule must be cyclic.
2. The molecule must be planar.
3. The molecule must have a complete conjugated π-electron system within the ring.
4. The molecule must have 4n π-electrons where n is any integer within the conjugated π-system.
This differs from aromaticity only in the fourth criterion: aromatic molecules have 4n +2 π-electrons in the conjugated π system and therefore follow Hückel’s rule. Non-aromatic molecules are either noncyclic, nonplanar, or do not have a complete conjugated π system within the ring.

In chemistry, an acid dissociation constant (also known as acidity constant, or acid-ionization constant; denoted Ka) is a quantitative measure of the strength of an acid in solution. It is the equilibrium constant for a chemical reaction.
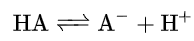
known as dissociation in the context of acid–base reactions. The chemical species HA is an acid that dissociates into A-, called the conjugate base of the acid, and a hydrogen ion, H+. The system is said to be in equilibrium when the concentrations of its components do not change over time, because both forward and backward reactions are occurring at the same rate.
The dissociation constant is defined by
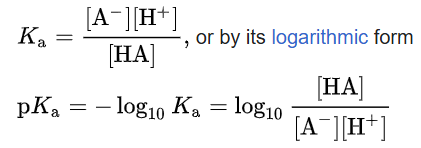
where quantities in square brackets represent the molar concentrations of the species at equilibrium. The form pKa is often used because it provides a convenient logarithmic scale, where a lower pKa corresponds to a stronger acid.