Correct option is A
Ans.(a)
No of atom in 12 g carbon
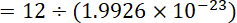
Thus Number of atoms in 1 mole carbon
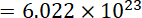
atoms
Ans.(a)
No of atom in 12 g carbon
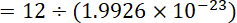
Thus Number of atoms in 1 mole carbon
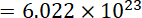
atoms
 English
English 200 Questions
200 Questions 200 Mins
200 Mins English
English English
English 200 Questions
200 Questions 200 Mins
200 Mins English
English

Suggested Test Series




Suggested Test Series



