Correct option is C
Ans.(c)
(A) In 3d-series, Manganese shows maximum number of oxidation states i.e., (+2, to +7)
(B) Zinc atom has completely filled d-orbitals in its ground state as well as in its oxidized state, thus it is not regarded as a transition element.
(C) Scandium shows only one oxidation state i.e., +3.
(D)
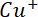
undergoes disproportionation reaction in aqueous solution
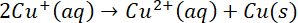





 English
English 200 Questions
200 Questions 200 Mins
200 Mins