Correct option is A
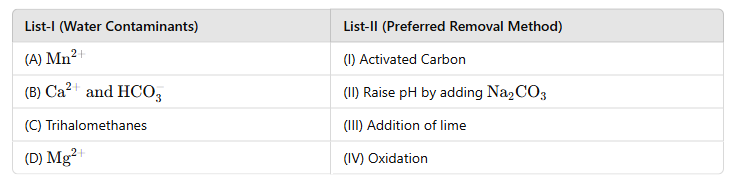
(A) Mn2+ → (IV) Oxidation:
·
Mn2+ ions are typically removed from water through oxidation, which converts manganese into its insoluble forms (e.g., manganese dioxide) that can be filtered out.
(B) Ca2+ HCO3− → (III) Addition of Lime:
· Hardness caused by
Ca2+ and
HCO3− can be treated by adding lime (Ca (OH)2) in a water softening process. This precipitates calcium carbonate and removes hardness.
(C) Trihalomethanes → (I) Activated Carbon:
· Trihalomethanes (THMs), which are disinfection byproducts, are effectively removed using activated carbon due to its adsorption properties.
(D) Mg2+ → (II) Raise pH by Adding Na2CO3:
· Mg2+ is removed by increasing the pH using
Na2CO3, which causes precipitation of magnesium hydroxide.
Information Booster: 1. Oxidation: Effective for removing metals like Mn2+ and Fe2+ from water.
2. Activated Carbon: Used for removing organic contaminants like THMs and odors from water.
3. Lime Addition: Removes temporary hardness caused by bicarbonates of calcium and magnesium.
4. Sodium Carbonate Addition: Precipitates magnesium as insoluble magnesium hydroxide by raising pH.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins

