Correct option is C
The reaction between methane (CH₄) and hydroxyl radical (OH•) follows second-order kinetics, which can be expressed as:
Rate=k×[CH4]×[OH•]
Where:
· k=8.36×10−21 m³ molecule⁻¹ s⁻¹ (rate constant)
· [CH4]=5.0×1019 molecules/m³
· [OH]=1.8×1012 molecules/m³
Now, substituting the values:
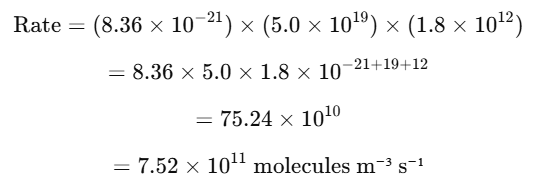
Additional Information:
· Second-order reactions depend on the concentration of both reactants, making their rate sensitive to changes in either.
· Methane (CH₄) oxidation by OH radicals is a key step in atmospheric chemistry, influencing the formation of greenhouse gases like CO₂.
· Hydroxyl radicals (OH•) are called "atmospheric detergents" as they help break down pollutants.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins

