Correct option is B
The temperature coefficient of a reaction is defined as the ratio of rate constants at two temperatures differing by 10K.
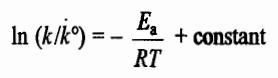
Ea: Activation energy
R: Gas constant
T: Absolute temperature

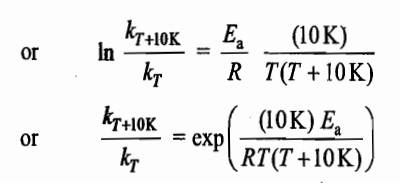
For many reactions at ordinary temperatures, the energy of activation is of the order of 80kJmol-1. Thus, at 300K, temperature coefficient of many reactions has a value of
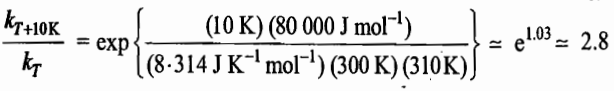
That is, the rate constant for the given value of Ea increases about 2.8 for every ten degree rise in temperature. For most reactions, the observed value usually lies between 2 and 3.





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

