Correct option is B
Infrared spectroscopy (IR spectroscopy or vibrational spectroscopy) is the measurement of the interaction of infrared radiation with matter by absorption, emission, or reflection. It is used to study and identify chemical substances or functional groups in solid, liquid, or gaseous forms. It can be used to characterize new materials or identify and verify known and unknown samples.
Theory
Infrared spectroscopy exploits the fact that molecules absorb frequencies that are characteristic of their structure. These absorptions occur at resonant frequencies, i.e. the frequency of the absorbed radiation matches the vibrational frequency. The energies are affected by the shape of the molecular potential energy surfaces, the masses of the atoms, and the associated vibronic coupling.
In particular, in the Born–Oppenheimer and harmonic approximations (i.e. when the molecular Hamiltonian corresponding to the electronic ground state can be approximated by a harmonic oscillator in the neighbourhood of the equilibrium molecular geometry), the resonant frequencies are associated with the normal modes of vibration corresponding to the molecular electronic ground state potential energy surface. Thus, it depends on both the nature of the bonds and the mass of the atoms that are involved. Using the Schrödinger equation leads to the selection rule for the vibrational quantum number in the system undergoing vibrational changes:
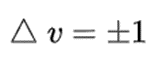
The compression and extension of a bond may be likened to the behaviour of a spring, but real molecules are hardly perfectly elastic in nature. If a bond between atoms is stretched, for instance, there comes a point at which the bond breaks and the molecule dissociates into atoms. Thus real molecules deviate from perfect harmonic motion and their molecular vibrational motion is anharmonic. An empirical expression that fits the energy curve of a diatomic molecule undergoing anharmonic extension and compression to a good approximation was derived by P.M. Morse, and is called the Morse function. Using the Schrödinger equation leads to the selection rule for the system undergoing vibrational changes:
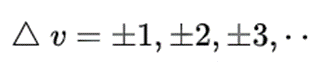
Number of vibrational modes
In order for a vibrational mode in a sample to be "IR active", it must be associated with changes in the molecular dipole moment. A permanent dipole is not necessary, as the rule requires only a change in dipole moment.
Asymmetrical diatomic molecules, e.g. carbon monoxide (CO), absorb in the IR spectrum. Heteronuclear diatomic molecules are indeed IR active. Heteronuclear diatomic molecules have different atoms bonded together, and they exhibit a chanqe in their dipole moment during vibration, which makes them IR active. Homonuclear diatomic molecules (those with identical atoms) do not show this behavior and are not typically IR-active.

Additional Knowledge
Statement-A
Infrared (IR) spectroscopy is not typically used to determine the band gap, band structure, or charge carrier concentration of a compound. These properties are more commonly studied using techniques like solid-state physics, electronic spectroscopy, and electrical measurements.






