Correct option is D
The complex [Cr(NH₃)₄(NO₂)₂]Cl can show both ionisation isomerism and linkage isomerism.
In ionisation isomerism, interchange of ligands takes place between coordination and the ionization sphere. They have the same molecular formula but give different ions in solution. Ionization isomerism can apply to metal complexes, ionisation isomerism relates to the way in which ligands get attached to the metal ion of these complexes. The importance of ionization isomerism is that it involves an exchange of ions inside and outside the coordination sphere. In ionization isomerism compounds give different ions in solution to the exchange of ions in the coordination sphere and the counter ion present in the complex.
For example:
· [Cr(NH₃) ₄(NO₂) ₂] Cl
· [Cr (NH₃) ₄(NO₂) Cl]NO₂
These two compounds are ionisation isomers, as they differ by the exchange of Cl⁻ and NO₂⁻ ions.
Linkage isomerism or ambidentate isomerism is a form of isomerism in which certain coordination compounds have the same composition but differ in their metal atom's connectivity to a ligand. Nitrite is an ambidentate ligand and can form a wide variety of coordination complexes by binding to metal ions in several ways.
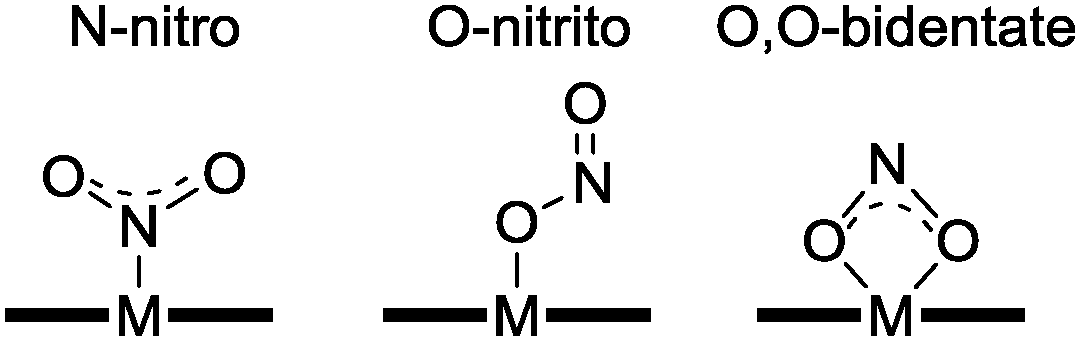
Additional Knowledge
Optical activity arises from chiral molecules-those that cannot be superimposed on their mirror images. This typically happens when the complex does not possess a plane of symmetry or a center of symmetry.
Geometric isomers are two or more coordination compounds which contain the same number and types of atoms, and bonds (i.e., the connectivity between atoms is the same), but which have different spatial arrangements of the atoms. The isomer in which like ligands are adjacent to one another is called the cis isomer. The isomer in which like ligands are opposite one another is called the trans isomer.
Both cis and trans geometry of the given complex leads to a plane of symmetry through the molecule, making it achiral.
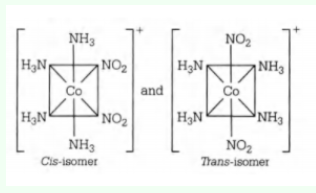





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

