Correct option is C
1. Statement (A):
· Correct.
· The BOD (Biochemical Oxygen Demand) test is indeed an indirect measure of organic matter. It quantifies the amount of oxygen required by microorganisms to decompose biodegradable organic matter in water.
2. Statement (B):
· Correct.
· The rate of DO (Dissolved Oxygen) consumption is directly proportional to the amount of biodegradable organic matter available at time ‘t’. This is based on first-order reaction kinetics.
3. Statement (C):
· Incorrect.
· The standard BOD curve follows first-order kinetics, not zero-order kinetics. The rate of oxygen demand decreases as the concentration of biodegradable material decreases.
4. Statement (D):
· Correct.
· For a specific type of waste with a defined rate constant, the ratio of ultimate BOD (BODu) and BOD5_55 is constant. The ultimate BOD is related to BODt via first-order reaction equations.
5. Statement (E):
· Correct.
· Wastes with the same BO00445 but different rate constants can have different ultimate BODs. A faster rate constant leads to more rapid oxygen demand, while a slower rate constant spreads the oxygen demand over a longer period.
Thus, the correct answer includes (A), (B), (D), and (E).
Information Booster: 1. BOD Overview:
· Definition: Biochemical Oxygen Demand is the amount of oxygen required by aerobic microorganisms to break down organic material in water.
· Measurement Period: Often measured over 5 days (BOD5) at 20°C.
2. Kinetics of BOD:
· Follows first-order reaction kinetics:
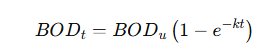
· BODt: BOD exerted at time t..
· BODu: Ultimate BOD.
· k: Reaction rate constant.
Applications of BOD Testing:
· Evaluating water quality in sewage and industrial effluents.
· Monitoring pollution levels in rivers and lakes.
Limitations of BOD Testing:
· Cannot account for non-biodegradable pollutants.
· Requires 5 days for standard results, making it less suitable for urgent analyses.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins

