Correct option is B
Ammonia acts as a ligand due to its lone pair of electrons on the nitrogen atom, which it can donate to the central metal ion like Cu2+. In an alkaline solution, ammonia exists as free molecules and can readily coordinate with Cu2+ to form [Cu(NH3)4]2+. However, in acidic solutions, excess H+ ions protonate ammonia, converting it into NH4+ , which lacks the lone pair necessary for coordination with Cu2+. This prevents the formation of the complex in acidic conditions.
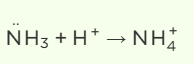
Information Booster
The stability of this complex arises from ammonia's strong ability to act as a Lewis base. In alkaline solutions, the lack of competing H+ ions allows ammonia molecules to effectively bind to the copper ion. The color change observed during this reaction is characteristic of the formation of this tetraammine complex, which is often a deep blue.
Additional Knowledge:
Hydration does protect metal ions, but it does not prevent ammonia coordination specifically. Hydration shells can be disrupted by strong ligands like ammonia, especially under alkaline conditions.
Copper hydroxide's amphoteric nature is irrelevant here. Amphoteric behavior relates to its ability to react with both acids and bases, not to complexation with ammonia.





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

