Correct option is D

A balloon containing an ideal gas is initially kept in an evacuated and insulated room. The balloon ruptures and the gas fills up the entire room. Which one of the following statements is true at the end of above process?

In a governor, what force exerted on the rotating flyweights controls the fuel consumption and also maintains a consistent engine speed?
Why does the velocity of fluid within the boundary layer start from zero at the surface and gradually increase to the free-stream velocity?
Which one of the following should be considered during a Brinell Hardness Test to accurately determine the hardness of a coarse material like a cast iron sample?
A traffic light is hanging from a horizontal pole that is supported by two cables attached to a vertical pole. The weight of the traffic light and the pole act downward. Given that the system is in equilibrium (the traffic light and the pole remain stationary) , which forces should be included in the Free Body Diagram?
In a certain language,
'Come To Home' is coded as 'Fa Ta Ha'
'Go To Home' is coded as 'Ta Ja Ha'
'Come For School' is coded as 'Fa Ya Ka'
Then what is the code for 'Come'?
What cold working technique is best suited for bending a long strip of iron metal into a roll shape?
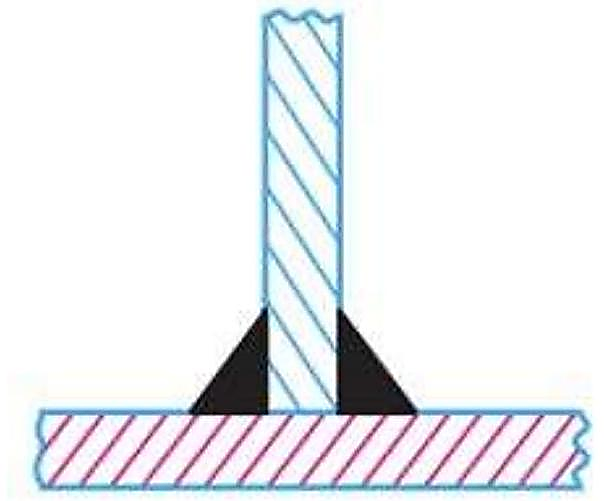
What type of joint is depicted in the image given above?
The base of a parallelogram is 10 cm and its height is 5 cm . What is the area of the parallelogram?
Which one of the following comparators is based on the principles of optics, using illumination, lenses, and mirrors to project a magnified silhouette of the part onto a viewing screen?
In turbomachinery, which of the following components form the sides of the triangle showing the relationships between the different velocity vectors of working fluid in a turbomachine?

